Ultrasonic Cleaning - Complete Guide
Ultrasonic cleaning using aqueous fluids is often used for final cleaning of precision components. It is gentle but very invasive and therefore good on intricate parts such as watches, valves, medical instruments and implants, circuit boards etc.
Ultrasonic action with water and detergants is suitable for most materials the exception being very soft metals i.e pure aluminium or very thin soft metals. The Ultrasonic action of cavitation will dent or erode these materials. If in doubt request cleaning trials first. For parts contaminated with thick grease etc a pre-wash is recommended before finally finishing with ultrasonic cleaning.
In some instances burnt on carbon ie. cylinder heads, injection moulding/plastic extrusion dies with burnt on residue can very effectively be cleaned with ultrasonic action and the correct combination of detergent.
Ultrasonic cleaning is based on 3 parameters:
THE MECHANICAL EFFECT:
How will ultrasonic waves help clean the parts?
THE CHEMICAL EFFECT:
Why is it important to use detergents?
Why can’t we clean only with water?
THE QUALITY OF WATER:
How to choose the best water quality?
How to control all of these parameters?
PROCESS CONTROL:
Check all parameters and get perfect cleaning
TIPS TO IMPROVE CLEANING EFFICIENCY:
What can you change?
THE MECHANICAL EFFECT - Ultrasonic Cleaning
How are ultrasonic waves produced?
When high frequency sound waves are passed through a cleaning fluid such as water with a suitable detergent additive, many millions of microscopic bubbles form and collapse. These bubbles are the result of the stretch and compress phrases of the sound waves within the fluid, the whole process is known as cavitation. Micro-bubbles will implode under the mechanical action of ultrasonic waves and a large amount of energy will be released and the local temperature will increase. The bubbles are forced into crevices where the fluid can penetrate between the contamination and the workpiece and leaving it totally clean and free of hidden soils.
The different ultrasonic frequencies
25 KHz - The most powerful frequency to remove strong pollution. Do NOT use on mirror polish surfaces and sensitive materials like glass, aluminum...
40 KHz - Standard frequency which can be compatible on a wide range of material and for removing a lot of pollution.
80 kHz - This frequency is used to clean parts with complex geometry. Cavitation bubbles can go into small holes to remove the pollution.
120 kHz and Megasonic - Main application in precision optics to clean very sensitive parts like wafers. The power of cavitation is low so these frequencies are used on clean parts to remove dust in final cleaning.
(80 kHz and above are only available on special request orders)
Important parameters which can influence the cavitation
- External pressure
- Temperature - Heated cleaning liquids reduce cleaning time and dirt is removed faster
- Ultrasound frequency
- Ultrasound power
- Nature of chemicals used to clean, its concentration, its density...
Positioning the parts in working chamber
The efficiency of water-based cleaning processes is related to the intimate contact of the cleaning chemistry and the impurities on the surface. The importance of positioning is always underestimated mainly with parts with complex geometry.
The performances with the right positioning of the parts in an ultrasonic cleaner are;
- Efficiency of the ultrasonic by dipping or pressure jet by spraying
- Physical protection from mechanical damage of the surface
- Efficiency of the contact between substrate and chemical specialties
- Efficiency of the rinsing operation
- Efficiency of the drying step
THE CHEMICAL EFFECT - Properties of the water molecule
Polarity of water
A water molecule is made of Oxygen and Hydrogen. Because of its intrinsic properties, the water molecule is polar which means water has the capacity to dissolve salts. But water is not miscible with non-polar hydrocarbons like oils and fats
Surface tension of a liquid
The paper clip is floating at the surface of the water, this is a force at which the surface is compensating for its weight resulting in intramolecular attraction (Van der Waals force) due to the polarity of the water molecule. Inside the liquid, the Van der Waals forces are compensating for each other. At the liquid’s surface, the resultant forces are going towards the inside of the liquid. This tends to contract the liquid and its surface behaves like a stretched membrane.

Lotus effect
Because of its high surface tension, natural water keeps its spherical form.
Corrosion effect - Iron Oxidation
This happens when Metallic iron dissolves in water by electron transfer. Dissolved oxygen reacts with water molecules and electrons to form hydoxyde ions. The Hydoxyde and iron ions then react to form iron hydroxyde. Various chemical combinations as oxydes or hydroxydes salts.
Why do we need to use detergents to clean parts?
- To decrease the surface tension of the water (wetting / wet effect)
- To allow solubilisation of oils and fats compounds in water solution (emulsifying effect)
- To protect against corrosion (adding corrosion inhibitors)
Steel wool 30 min in water

THE CHEMICAL EFFECT - How do detergents work?
DEGREASING – Nature of oil and fat
Fats from plant and animal origin saponification in caustic solution.
- Triglyceride is not soluble in water
- Carboxylate ion and glycerol are water soluble
- Pay attention to the substrate sensitivity towards strong alcaline solution (tarnishing of copper alloy)
Fats from mineral origin saponification is chemically not possible Tenside effect is requested
What is a surfactant?
A surfactant is composed of a hydrophobic tail and a hydrophilic head. The length of the hydrophobic tail and the nature of the head will induce the ability to create micelles and the proper action of detergency.
How do surfactants work?
The Critical Micelle Concentration (CMC): The minimum concentration at which surfactants molecules begin to form micelles. Without micelles, no cleaning properties can be achieved by the detergent. Below the CMC, the molecules are free and form monomolecular layers (to adsorb). Above the CMC, they form the monomolecular layer. When all the area has been covered by surfactants, they form the micelles. More surfactants are added, more micelles are formed. The surface tension does not decrease with the increasing of surfactants.
Decrease water surface tension: The surfactants orientate themselves in between the water molecules and disturb their “hand-holding”. The force is then lowered. For some surfactants the surface tension can be reduced to ~30 mN /m or even lower.
Emulsify oils: When oil is added to water containing a surfactant, oil is surrounded by surfactant. The surfactant head is bonded with water molecules while the tail catches the oil droplets.

Disperse oils into the liquid: Surfactants adsorbing onto the soil/ oil droplets
- Hydrophilic group be in contact with aqueous phase
- Hydrophobic group in contact with oil or soil
Stabilization of the droplets helps prevent it from coalescence with other droplets (by repulsive forces). Repulsion forces between the head Groups avoid coalescence of the droplets.
Specific effect - corrosion inhibition: Monomolecular film on a solid surface Hydrophobic effect Protection against oxidation
There are four families of surfactants
- Anionic: for the wettability
- Cationic: for antistatic properties and antibacterial activities
- Amphoteric: surfactant will be anionic or cationic as a function of the pH value of the solution
- Nonionic: for emulsifying effect
The effects from surfactants; decrease the surface tension of the water, emulsifies the pollution, disperses the pollution and protects against corrosion.

Typical composition of detergent:
- Surfactants
- Sequestering agents: components for inhibition of negative calcium and magnesium salt effects (reaction with surfactants and low soluble salts deposition)
- Solvents
- pH adjustment additives
- Anti-corrosive additives
- Specific additives: preservatives, biocides, anti-static agents, thickeners...
- Fillers
Three parameters will induce the properties of detergent:
The substrate - Product has to be compatible with the substrate. Its pH value and composition has to be adapted.
The pollution - Product has to be efficient on specific pollution.
The surface treatment of the substrate -What treatment will be applied on the parts after cleaning?
Metallic substrates
| Metal | Behaviors | Observations | |
|---|---|---|---|
| Alkaline medium | Acidic medium | ||
| Stainless Steel | Resistant excepted in presence of chlorides | Resistant excepted to chlorhydric acid and chlorides | Nitric acid and phosphoric acid will create a passivation layer onto the material. Chlorides will pickle the protective layer. |
| Casting iron | Resistant | Sensitive to corrosion | Phosphoric acid will create a passivation layer. |
| Copper-Brass-Bronze | Corrosion by colouration change | Sensitive to corrosion | Strong etching with nitric acid. More or less sensitive to ammonium salts |
| Aluminum-Zinc and alloys | Sensitive to corrosion | Resistant | White corrosion and tarnish effect can appear |
| Nickel | Resistant | ||
| Chrome | Resistant | ||
| Titanium alloys | Stains can appear | Acidic medium are used as passivation agent | |
| Gold | Resistant | Resistant | |
Non metallic substrates
Ceramics - Sensitivity is a function of the composition and the surface statement of the material.
Mineral glasses in ophthalmic optics - Resistant to chemical compounds
Sapphires - Resistant to chemical compounds
Mineral glasses in precision optics - Sensitive to alkaline, acidic products and to phosphates.
Organic lenses in ophthalmic optics (CR39, PA, High index) - Low sensitivity
Organic lenses in ophthalmic optics (polycarbonates) - Sensitive to alkaline products and specific solvents
IMPORTANT: Take into account sensitivity: To the corrosion effect due to the pH and the conductivity of water. To sequestering agents which can solubilize some elements of materials and create holes.
One specific example: the sensitivity of tungsten carbides bonded with cobalt. Carbide grains are included in a cobalt matrix. The cobalt can be leached quickly by an aqueous cleaning solution. The cobalt leaching will have a strong negative impact on the mechanical properties of the material.
The pollution
Polishing compounds - Animal and vegetal fats for grinding and polishing mineral content
Oils and emulsions - Mineral and vegetal oils; water-based emulsion
Protection lacquers - Organic layer for mechanical protection during manufacturing processes
Oxidation residues - On copper alloys or sensitive steel (20 AP) during manufacturing processes or storage
Tumbling residues - For surface finishing after mechano-chemical polishing with abrasive stones in tumbler
Finger prints - After latest step of manufacturing process and before controlling
Surface treatment of substrate
According to the next steps after cleaning, the surface of substrate must be hydrophilic or hydrophobic.
The wettability can be evaluated by measuring the contact angle. The droplet will form an angle with the substrate:
- If θ > 90° the surface is non-wetting
- If θ < 90° the surface is partial wetting

Hydrophilic surface will be required for final cleaning to obtain a surface without any particles. For Adhesion of specific coatings with polar or hydrophilic behavior and for helping to rinse the parts.
Hydrophobic surface will be required for providing corrosion protection, getting an antistatic behavior, adhesion of specific coatings with non polar or hydrophobic behavior and for helping to dry the parts.

View our full range of ultrasonic cleaning fluids here
WATER QUALITY INFLUENCE - Rinsing stages
Typical rinsing steps
- Tap water rinse for a high rinsing effect. (tap water has a heterogeneous composition depending on its supply source)
- Di water for spot-free drying. (Di water has a standardized chemical quality)
Water qualities
TAP WATER
- Preparation of cleaning solution and first rinsing step when
- Water quality is homogeneous and no critical surface quality is needed
- Very heterogeneous quality, depending on site
- High conductivity up to 600-700 microSiemens / cm
- Often high content of calcium and magnesium salts
- which leads to salt residues on the parts
- May be contaminated by organic and chlorine materials
SOFTENED TAP WATER
- First rinsing step when tap water has a very high calcium and magnesium content
- Similar quality and conductivity as the original tap water
- But exclusion of calcium and magnesium replaced by sodium (ion exchanger)
- Inhibition of low soluble salt deposition
- May be contaminated with bacteria

OSMOSIS WATER
- Preparation of cleaning solution when tap water has a very high calcium and magnesium content
- Produced by low porosity membrane filtration
- 90% salt retention; conductivity: 10-20 microSiemens / cm
- Elimination of most organic and bacterial contamination
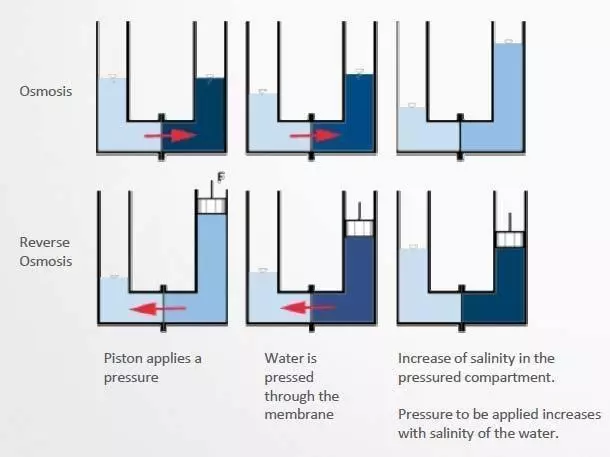
DEMINERALIZED WATER
- Last rinsing step before drying for total elimination of residual salts
- Produced and recycled through ion exchangers (mixed bed resins)
- Very low salt content; conductivity: < 0.1 microSiemens / cm
- May be contaminated with organics and bacteria
- need for UV and active charcoal complementary treatment
Ions exchanger is a solid material insoluble in water (small balls). The material is chemically grafted with ionic functions able to fix ionic species like dissolved salts.
R-AR + + BS+ = R-BR+ + AS+
R-AR - + BS- = R-BR- + AS-
Cationic ions exchangers retains cations like Na+ Anionic ions exchangers retains anions like Cl-. A mixture of cationic and anionic ions exchangers is a mixed bed
WATER QUALITY INFLUENCE - Microbiology
Critical parameter of process often forgotten
Bacteria/algae will grow in an aqueous medium (humidity) and at temperatures between 20°C to 40°C. They will appear in the demineralized water plant and also in the rinsing tank. (If tank walls are slippery, then there are bacteria.) IMPORTANT: BACTERIA and ALGAE can also appear in cleaning solutions if product has a neutral pH value and it is used at low temperatures.
PROCESS CONTROL - Cleaning solution
Due to the internal attractive forces of a liquid, air bubbles within the liquids are compressed. The resulting pressure (bubble pressure) rises at a decreasing bubble radius. The bubble pressure method makes use of this bubble pressure which is higher than in the surrounding environment (water). A gas stream is pumped into a capillary that is immersed in a fluid. The resulting bubble at the end of the capillary tip continually becomes bigger in surface, while the bubble radius decreases.
The pressure rises to a maximum level. At this point the bubble has achieved its smallest radius (the capillary radius) and begins to form a hemisphere. Beyond this point the bubble quickly increases in size and soon bursts, tearing away from the capillary, thereby allowing a new bubble to develop at the capillary tip. It is during this process that a characteristic pressure pattern develops (see picture), which is evaluated for determining the surface tension.
Cleaning test equipment
The refraction index measurement
The principle of operation is based on the total reflection of an incident beam at the boundary of the sample and the glass prisms between which it is sandwiched. This necessitates that the sample has a lower refractive index than the prism, so prisms are made of high-index glass.
For one compound the refraction index will be a function of its concentration and the temperature. If temperature is fixed, the standard curve of the refraction index as a function of concentration can be established.

UPC 3000 (Ultrasonic Process Controller)
Simple and immediate monitoring of key parameters to insure the quality of cleaning
- Direct measurement of temperature and conductivity
- Monitoring of the ultrasonic power variations in the baths
- Quality control of the DI-Water in the final rinsing tanks
- Specific configuration of the device based on customer needs
- Control of the device through use of a simple menu and three keys
- Measurement of the concentration of detergent retrieved in hard water or DI-Water
AQUASNAP
- The system measures ATP in bacteria.
- Each bacteria is composed of a fixed amount of ATP.
- Snap: liquid and reagents are mixed, the ATP binds to a reagent which emits light (luciferase / luciferin).
- The amount of light is measured by the device (unit: Relative Light Unit).
- The amount of ATP is back-calculated and the amount of bacteria can be deduced.

Dyne test pens
Determination of surfaces tension with inks: This testing method is well suitable for the operating people on production lines as a routine check. The result can be immediately evaluated and gives a very clear picture of the degree of treatment or cleaning to accordingly trained staff. The marking inks show that the surface tension of this surface is at least 30-32 mN/m.

Contact angles measurements
To determine surface energy, contact angle between surface and liquid drop is needed. This is possible with a picture of drop shape over material we want to characterize. If the liquid is water:
- Big angle means an hydrophobic surface
- Small angle means an hydrophilic surface

Tips to improve cleaning efficiency
Aqueous process (water represents up to 95%!).
Stabilize your water quality
Filtration cartridges
- Only for particles with a porosity between 5µm and 20µm
- Good rinsing effect
- Dissolved salt not filtered
- Similar conductivity before and after filtration
- Bacteria and chlorine not retained
Softening Device
- Dissolved salt retained
- Similar conductivity before and after filtration
- Reduced rinsing effect (Ca2+ and Mg2+ replaced by Na+)
- Bacteria and chlorine not retained
Mix of technologies
- Retains particles in suspension (porosity to be adapted)
- Keeps a good rinsing effect
- Retains bacteria and chlorine
Filtration Cartridge + Softening + Nano-filtration necessary
Stabilize your process
Stable processes and a constant lens surface quality need control. Key parameters to be controlled:
1/ CONCENTRATION OF ACTIVE TANKS
- How can I control the concentration?
- How do I set up & control the frequency?
- Refresh?
- Change?
2/ ULTRASONIC POWER IN EACH TANK
- Are my transducers efficient?
- Is my Ultrasonic power sufficient (equipment defect) ?
3/ CONDUCTIVITY VALUE IN EACH ACTIVE TANK
- Is my conductivity in compliance with the supplier?
- Correlation between conductivity and pollution?
4/ CONDUCTIVITY VALUE IN THE DI-WATER TANK
- Does the conductivity in the tank match the DI-Unit measurement?
- Is my DI-Water quality good enough?
CONDUCTIVITY? Electric conductivity is the reverse of the resistivity. It corresponds to the conductance of a material portion of 1 m length and 1 m2 of section.






